Therapist
Berezhko
Olga Igorevna
35 years of experience
General practitioner of the highest category, member of the Russian Scientific Medical Society of Therapists
Make an appointment
Vasculitis is a group of vascular diseases that are characterized by the development of an inflammatory process and subsequent necrosis of the vascular walls. The disease is also called arteritis or angiitis. As the disease progresses, a significant deterioration in blood circulation occurs. Pathology can affect arteries, veins, capillaries, venules and arterioles.
Types and classification of vasculitis
Vasculitis is divided into types:
- primary;
- secondary;
- systemic.
In the first case, we are talking about an independent and still just beginning disease. The second type may appear with other pathologies, for example, tumors. It can also occur as a reaction to an infection. Systemic vasculitis can occur in different ways. It leads to inflammation of blood vessels and damage to their walls, and tissue necrosis cannot be ruled out. Without proper treatment, complications usually occur, including death.
Based on the caliber of the affected vessels, a classification of vasculitis was identified. It may be in the form:
- arteritis (affects large vessels);
- phlebitis (affects the walls of the veins);
- arteriolitis (inflames the arteries and arterioles);
- capillaritis (affects capillaries).
Also included in a separate category is vasculopathy, which is characterized by the absence of clear signs of inflammation and infiltration of the vascular walls.
The degree of vascular damage can be mild, moderate and severe.
Main primary vasculitis:
- Takayasu arteritis (synonyms: “nonspecific aortoarteritis”,
- "aortic arch syndrome")
- giant cell arteritis (synonyms: “temporal arteritis”,
- "senile arteritis", "Horton's disease")
- polyarteritis nodosa
- Kawasaki disease
- granulomatosis with polyangiitis (synonym: “Wegener’s granulomatosis”)
- microscopic polyangiitis
- eosinophilic granulomatosis with polyangiitis (synonym: “Churg-Strauss syndrome”)
- cryoglobulinemic vasculitis
- IgA vasculitis (synonyms: “hemorrhagic vasculitis”, “Henoch-Schönlein purpura”)
- Hypocomplementary urticarial vasculitis (synonym: “anti-C1q-associated vasculitis”)
- Anti-BMK-associated disease (synonym: “Goodpasture syndrome”)
Primary vasculitis is an independent disease that falls primarily within the competence of rheumatologists, although the diagnosis and treatment of these diseases is impossible without the participation of doctors of other specialties: otolaryngologists, ophthalmologists, neurologists, dermatologists, etc.
The caliber of affected blood vessels in different forms of vasculitis is not the same and varies from large arteries (with a diameter of 1.0 cm or more) to the smallest arterioles, capillaries and venules, visible only under a microscope. The type of inflammation is also different. All this explains the extremely pronounced diversity of clinical manifestations of primary systemic vasculitis and their dissimilarity from each other.
Causes of the disease
The reasons for the formation of primary vasculitis are still unknown to science. Secondary disease can occur against the background of:
- acute or chronic infection;
- genetic (hereditary) predisposition;
- thermal effects (overheating or hypothermia of the body);
- various injuries;
- burns;
- contact with chemicals and poisons.
Another cause of vasculitis can be viral hepatitis. One or more risk factors are enough to change the antigenic structure of tissues. In this case, the immune system begins to secrete antibodies, which further damage the tissue, in this case the blood vessels. This leads to an autoimmune reaction and inflammatory and degenerative processes.
Symptoms of vasculitis
Regardless of the type and form of the disease, the symptoms of vasculitis are similar. These include:
- decreased body temperature;
- joint pain;
- pale skin;
- regularly recurring sinusitis;
- vomiting;
- nausea;
- myalgia;
- rash in the form of spots, urticaria, subcutaneous nodules, hemorrhagic purpura, ulcers or blisters;
- sudden mood swings;
- convulsions;
- intoxication;
- drowsiness;
- sometimes pleurisy, bronchial asthma;
- loss of sensation (insemination);
- exacerbation of cardiovascular diseases;
- weakness;
- lack of appetite followed by weight loss.
Depending on the location, vasculitis can also manifest itself through a sharp deterioration in vision, inflammation and swelling of the eyes, pain in the lower back, in the kidney area, abdominal cramps and disruption of the gastrointestinal tract.
Are you experiencing symptoms of vasculitis?
Only a doctor can accurately diagnose the disease. Don't delay your consultation - call
Cutaneous vasculitides comprise a broad heterogeneous group of diseases affecting the blood vessels of the skin and are clinically characterized by polymorphous skin lesions, including palpable purpura, as well as urticarial and necrotizing ulcerative lesions with possible, although rare, extracutaneous involvement. Cutaneous leukocytoclastic angiitis and urticarial vasculitis, which are the two prototypical and most common variations of this group, are usually idiopathic but may also be induced by various triggers, particularly drugs and infections, or may occur in association with systemic disorders, especially lupus erythematosus.
Adverse effects of vasculitis may also occur during relapse of chronic systemic vasculitis, such as granulomatosis with polyangitis and eosinophilic granulomatosis with polyangitis, with which cutaneous vasculitides may share clinical and histological features. To make a diagnosis of a specific form of vasculitis, the patient's skin reaction and clinical history must be compared with the histologic pattern, direct immunofluorescence results, laboratory findings, and the possible presence of extracutaneous manifestations. Histology is critical for the diagnosis of vasculitis, and the timing of biopsy is relevant to the correct assessment of the characteristics of the inflammatory infiltrate. In this review, we will focus in particular on the histological features of cutaneous vasculitides to distinguish them from systemic forms and to avoid misdiagnosis when the cutaneous manifestation is a feature of multisystem vasculitis.
Vasculitides are a broad group of diseases characterized histologically by an inflammatory infiltrate in/or around vessels and their damage, recognized as fibrinoid necrosis, which serves as a key to its diagnosis, as well as leukocytoclasia, thrombi, or connective tissue degeneration. Vasculitis may be an underlying disorder or a condition secondary to systemic diseases such as connective tissue diseases, malignancies, or a consequence of drug use. Skin lesions are very common in vasculitis and can be classified as vasculitis of a single organ or part of a multi-organ disease in which the skin is the site of onset of the disease. Clinical presentation depends on the type and caliber of vessel involved, showing purpuric macules and infiltrated erythema when only the superficial dermal vessels are involved, or a polymorphic appearance with palpable purpura, bullous lesions, ulcers and nodules when the deeper dermis and subcutaneous tissue are also involved.
From a histological point of view, most cutaneous vasculitides affect small vessels, especially venules, and are characterized by an acute neutrophilic inflammatory infiltrate that characterizes the features of leukocytoclastic vasculitis (LCV). To achieve a correct diagnosis, it is necessary to integrate the manifestation of LCV with the type of vessel (small and/or muscular), the underlying inflammation, extravascular manifestations such as tissue eosinophilia, tissue neutrophilia or granulomas, and direct immunofluorescence (DIF) findings.
However, the histological pattern of skin lesions is often the same in vasculitis limited to the skin, such as cutaneous leukocytoclastic angiitis (CLA), as well as in systemic vasculitis, such as granulomatosis with polyangititis (GPA) or eosinophilic granulomatosis with polyangititis (EGPA), since the latter may lack granulomatous aspects in the affected skin. Histology should be correlated with clinical findings such as systemic symptoms and signs, instrumental tests potentially revealing visceral involvement, and laboratory parameters such as routine blood count, urine count, and antineutrophil cytoplasmic antibody (ANCA).
Thus, taking into account the relationship between clinical parameters and histology, we will focus on the histological presentation of cutaneous vasculitides, as well as the histopathological features of the cutaneous manifestations that occur during the main forms of systemic vasculitis.
Classification of vasculitis
The classification of vasculitides has been challenging and much debated in the last half of the century. The nomenclature adopted by the 2012 Chapel Hill International Consensus Conference (CHCC) is the most widely used, although some points of contention continue to be debated (Table I).
Compared to the previous classification, some traditional eponymous terms have been excluded, such as Wegener's granulomatosis and Churg-Strauss syndrome. Additionally, vasculitides are classified based on the size of the vessels affected and the pathogenesis, in addition to the main organ affected. Cutaneous vasculitides have been divided into two groups: small vessel vasculitis (SVV) and segmental vasculitis (SOV). SVVs included vasculitides that predominantly affect small vessels (intraparechal arteries, arterioles, capillaries, and venules) but rarely affect medium-sized arteries and veins. SOV are vasculitis affecting the vessels of one organ without evidence of systemic involvement. In 2012, the International Consensus Conference did not include two types of vasculitis limited to the skin, persistent elevated erythema and acute hemorrhagic edema of the newborn.
How to Perform a Skin Biopsy
The location, depth and timing of the biopsy are three key factors for diagnosis. A superficial dermal biopsy (such as a punch biopsy) is performed for macular or purpuric skin lesions. Deeper incisional biopsies, which involve sampling subcutaneous fat, are performed in the presence of nodules or lesions of medium-sized vessels, such as polyarteritis nodosa (PAN).
The timing of the biopsy is an important factor because vasculitis is a dynamic process and, regardless of its cause, within the first 24 hours neutrophils penetrate the vessel wall with fibrin deposition, which leads to the formation of an acute inflammatory infiltrate and vascular damage; after 24 hours, neutrophils begin to be replaced by lymphocytes and macrophages; after 48 hours there is usually progression to a chronic lymphocyte-rich infiltrate with neovascularization.
For these reasons, a biopsy should be performed when the lesion is new, approximately 24-48 hours later. Timing is also important for DIF, as immunoglobulins (Igs) are intensely positive during the first 48 hours, but after 72 hours inflammation destroys the immune complexes and the lesions become false negative. Complement can be detected in >50% of lesions at 72 hours.
Table I. Nomenclature of systemic vasculitides (Chapel Hill Consensus Conference 2012)
Nomenclature of vasculitis Large vascular vasculitis (LVV) - Takayasu arteritis (TAK) - Giant cell arteritis (GCA) Medium vascular vasculitis (MVV) - Polyarteritis nodosa (PAN) - Kawasaki disease (KD) Small vascular vasculitis (SVV) - ANCA - associated vasculitis (ANCA) - Microscopic polyangiitis (MPA) - Granulomatosis with polyangiitis (Wegener's) (GPA) - Eosinophilic granulomatosis with polyangiitis (Chardg-Strauss) (EGPA) - Immune complex vasculitides (SVV) - Antiglomerular basement membrane antibody disease (anti - GBM) - Cryoglobulinemic vasculitis (CV) - IgA vasculitis (Henoch-Schönlein purpura) (IgAV) - Hypocomplementemic urticarial vasculitis (anti - C1q) (HUV) Various vascular vasculitis (VVV) - Behçet's disease (BD) - Cogan's syndrome (CS) Single organ vasculitides (SOV) - Skin leukocyte antigen - Cutaneous arteritis - CNS-predominant vasculitis - Isolated aortitis - Other Vasculitides associated with systemic diseases - Lupus vasculitis - Rheumatoid vasculitis - Sarcoid vasculitis - Other Vasculitides associated with probable pathology - Cryoglobulinemic vasculitis associated with hepatitis C virus - Vasculitis associated with hepatitis B virus - Aortitis associated with syphilis - Immune complex drug-associated vasculitis - ANCA - drug-associated vasculitis - Vasculitis associated with cancer - Others
Cutaneous vasculitis: histological features
Skin biopsy is the gold standard for cutaneous vasculitis. Firstly, to distinguish it from other diseases that present a similar clinical picture. Cutaneous pseudovasculitides are a heterogeneous group of diseases that may be characterized by hemorrhagic lesions due to vessel wall dysfunction (eg, amyloidosis, scurvy, pigmented purpuric dermatitis, and coagulation disorders) or cyanotic and ulcerative lesions due to vascular occlusion (eg, purpura fulminans, warfarin necrosis, antiphospholipid syndrome, cardiac fibroids, cholesterol embolization and calciphylaxis). Diagnosis of vasculitis is based on evidence of an angiocentric and/or angioinvasive inflammatory infiltrate accompanied by fibrin deposition within and around the vessel (also known as fibrinoid necrosis), and hematoxylin and eosin staining of newly affected skin areas. Other common features, but not exclusive to vasculitis, are extravasation of red blood cells, perivascular “nuclear dust,” and signs of endothelial damage with endothelial swelling, desquamation, and necrosis. The second step in classifying and differentiating the type of vasculitis determines the size and distribution (ie, superficial dermis or subcutaneous tissue) and predominant inflammatory cells of the perivascular infiltrate. In addition, extravascular findings should be considered to improve the differential diagnosis, such as tissue eosinophilia (in EGPA), the presence of “red” granulomas (in GPA), or a combination of histopathological features consistent with the underlying disorder in connective tissue-associated vasculitides. The presence of pathophysiological markers such as DIF immune complex deposition or ANCA positivity should be integrated to establish a differential diagnosis of skin-limited versus systemic disease. For example, neutrophilic small superficial vessel vasculitis can be a histologic feature of both CLA and Henoch–Schönlein purpura (HSP), but the presence of DIF in IgA supports the diagnosis of the latter disease.
Cutaneous leukocytoclastic vasculitis
Leukocytoclastic vasculitis (LCV) is the typical histological pattern of most cutaneous vasculitides and is not a diagnosis in itself, although it is often used in place of CLA. This is an acute form of vasculitis, histologically characterized by an infiltrate of predominantly neutrophils, localized in most cases around small venules in the upper dermis; less commonly, the infiltrate affects the deep cutaneous and hypodermal small vessels and/or muscle vessels. “Nuclear dust” and extravasation of red blood cells are often present, and signs of thrombi are rarely detected.
Cutaneous leukocytoclastic angiitis
CLA is a postcapillary venule vasculitis caused by an immune complex reaction. It is often idiopathic but can be caused by medications or infections. CLA is a diagnosis by exclusion because, by definition, systemic disease must be absent. The disease usually affects the health of middle-aged adults (more women than men).
Macroscopy
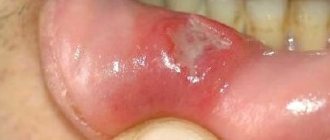
Clinically, it is characterized by self-limited petechial lesions or palpable purpura on the lower extremities (Figure 1A), sometimes extending to the buttocks, lower trunk, and shoulders. Ulcerative infections may progress (Figure 1B), healing with postinflammatory hyperpigmentation. The lesions usually resolve within a few weeks or months, but 10% of patients may experience a relapse lasting several years.
Microscopy
Characterized by typical features of leukocytoclastic vasculitis (Figure 1C,D). The presence of tissue eosinophilia may be associated with drug etiology. Figure 1. Cutaneous leukocytoclastic angiitis. Polymorphous skin manifestations including palpable purpura (A) and necrotizing ulcers associated with purpuric lesions (B) on the lower extremities. Typical histologic features of leukocytoclastic vasculitis with sparse neutrophil infiltrate and nuclear debris localized around dermal venules (C, D).
Auxiliary tests
DIF of early lesions reveals perivascular deposits of IgG, IgM, and/or complement in a granular specimen. Fibrin deposits can be observed in early and late lesions.
Henoch-Schönlein purpura
It is a vascular vasculitis caused by the deposition of IgA immune complexes. This is common in children during the winter months, but rare in adults. In approximately 30% of cases, the trigger is an upper respiratory tract infection, especially Staphylococcus aureus; less commonly, HSP is caused by medications. In addition to the skin, which is sometimes the only symptom of the disease, HSP can affect the gastrointestinal system, kidneys and joints.
Macroscopy
Palpable hemorrhagic purpura is symmetrically localized on the limbs and buttocks. Rare localization to the trunk and face, as well as evidence of clinical edema, appear to be associated with gastrointestinal involvement. Ischemic and necrotic lesions are possible. The rash is usually self-limiting after a few days, but may recur over many years. Skin lesions may be associated or accompanied by arthralgia (in 75% of patients), abdominal pain, sometimes with gastrointestinal bleeding (in 65% of cases) and nephritis (in 40% of cases) with microscopic or macroscopic hematuria and proteinuria. The prognosis is usually good in cases with mild renal impairment and neurological symptoms.
Microscopy
It is characterized by typical features of leukocytoclastic vasculitis with extravasation of red blood cells. The presence of papillary cutaneous edema is apparently associated with renal failure.
Auxiliary tests
IgA deposits, mainly of the IgA1 subclass, are visible on the vessel walls of affected or unaffected skin on DIF.
Urticarial vasculitis
Urticarial vasculitis (UV) is a chronic, relapsing disease characterized by blistering that persists for more than 24 hours. Based on the levels of complement proteins in the blood, UV can be classified as hypocomplementary (HUV), which often occurs with systemic involvement, especially renal failure, and normocomplementary (NUV), which usually occurs without systemic manifestations. HUV is associated with a reduction in serum complement (C)1q due to complement activation by immune complexes formed between IgG autoantibodies and the collagen-like region of C1q. In most cases, UV is idiopathic, but occurs in association with chronic infections (hepatitis B and C), hematological diseases (myeloma, IgM gammopathy, Castleman's disease, polycythemia vera), exposure to ultraviolet light and cold, exercise, systemic lupus erythematosus ( SLE), Sjögren's syndrome and malignant tumors.
Figure 2. A) Large blisters on the body of a patient affected by urtical vasculitis; B) leukocytoclastic vasculitis with a rare infiltrate of neutrophils and eosinophils in the dermis and around the venules, with upper cutaneous edema.
Macroscopy
Characterized by the appearance of blistering papules and plaques (Fig. 2A). UV skin lesions differ from regular urticaria in that they are painful and “burn” rather than “itch”, and persist for more than 24 hours and result in bruising and hyperpigmentation. The clinical course is chronic with unpredictable relapses over 3-4 years. Angioedema may be present, as well as purpura, livedo, or other skin lesions characteristic of CLA (papules, nodules, or necrotic-ulcerative lesions). HUV may be associated with systemic symptoms such as fever, malaise, myalgia and arthralgia, and may also involve organs (glomerulonephritis, abdominal pain and diarrhea, dyspnea and ophthalmic complications).
Microscopy
UV presents as a sparse or dense perivascular neutrophilic infiltrate with leukocytoclasia, usually without fibrin deposits. Tissue eosinophilia is more common in NUVs. Upper cutaneous edema may be present (Figure 2B).
Auxiliary tests
DIF detects perivascular deposits of IgM and/or C3 in the upper dermis, but is often negative in NUV. In HUVs, plasma levels of C1q, C3 and C4 are low, and circulating C1q and antinuclear autoantibodies can be detected. IgG and C3 at the dermoepidermal junction indicate a positive lupus test may be related to HUV with underlying SLE.
Cryoglobulinemic vasculitis
Cryoglobulinemic vasculitis (CV) is a rare small/medium vessel vasculitis caused by the deposition of cryoglobulins in the vessels. Cryoglobulins are cold-soluble immune complexes of rheumatoid factor (RF), Igs (IgM and less commonly IgG and IgA) and often HCV RNA. They precipitate and deposit on the vascular endothelium when the serum is cooled and are redissolved when the serum is warmed. CV may manifest as type I cryoglobulinemia, characterized by monoclonal IgM, associated with lymphoproliferative disorders (lymphoma, myeloma, Waldenström's macroglobulinemia, chronic lymphocytic leukemia) or as mixed cryoglobulinemia type II (monoclonal and polyclonal IgM and IgG) and type III (polyclonal IgM/IgG) ), which is commonly associated with hepatitis, rheumatologic disorders, and Epstein-Barr virus. However, CV is idiopathic in 50% of cases. Cutaneous manifestations are often associated with arthralgia, weakness, fever, weight loss, polyneuropathy, and renal damage.
Macroscopy
The classic clinical presentation is nonpruritic palpable purpura, urticarial papules, livedo reticularis, acrocyanosis, and painful ulcers on the lower extremities. The lesions develop over 3–10 days with hyperpigmentation, and the course usually recurs chronically over several months or years. Rarely, CV can occur on the upper extremities, oral and nasal mucous membranes. Other skin manifestations may also occur, such as urticaria, ulcers, plaques, acrocyanosis and cyanosis.
Microscopy
Leukocytoclastic vasculitis of postcapillary venules with extravasation of red blood cells is usually observed in CV occurring during mixed cryoglobulinemia types II and III. The histological characteristics of skin lesions in cryoglobulinemia type I are not true vasculitis, but are characterized by the presence of a perivascular infiltrate of predominantly mononuclear cells, thrombi, deposited cryoglobulins in the dermal blood vessels and extravasated red blood cells.
Supporting research
DIF shows deposits of IgM and complement in the vessel wall. Cryoglobulins are found in peripheral blood. Typically, ANCA are negative and hypocomplexity is present. High RF titer in 70% of cases.
Rheumatoid vasculitis
Twelve percent of cutaneous vasculitis cases are associated with connective tissue disease (CTD), and the most common and best defined are SLE with lupus vasculitis (LV) and rheumatoid vasculitis (RV). In SLE, cutaneous vasculitis occurs in approximately 19% of patients, representing nonspecific lesions based on the modified Gilliam classification. RV is probably the most widely recognized secondary vasculitis.
Macroscopy
Cutaneous manifestations are numerous and include palpable purpura, vesicomuscular and urticarial lesions, gangrene, skin ulcers, and nail bed infarction.
Microscopy
Cutaneous LV is usually characterized by a mild neutrophilic vasculitis, and less commonly by a neutrophilic muscular vascular vasculitis (similar to polyarteritis). Extravascular pallosed neutrophils and granulomas are possible. The presence of histological features of cutaneous lupus erythematosus is useful for diagnosis. In RV, the histological picture is characterized by LCV involving small blood vessels in the superficial and middle dermis and, less commonly, involving larger vessels in the superficial and deeper dermis.
Supporting research
Vascular deposition of IgM and C3 is widespread in RV. In the LV, a lupus test is often performed, characterized by IgG, IgM, and C3 deposits at the dermoepidermal junction. Atypical ANCA (a-ANCA) may be positive.
Vasculitis with granulomatosis
Another histological pattern of vasculitis is characterized by the presence of variable degrees of granulomatosis, both angiocentric and nonvascular, but related to vasculitis.
Granulomatosis with polyangitis (Wegener's granulomatosis)
Granulomatosis with polyangititis (GPA) is a rare immunologically determined multisystem disease characterized by the presence of granulomatous inflammation of the upper and/or lower respiratory tract, glomerulonephritis and systemic necrotizing vasculitis of small and medium vessels. It usually develops in the fourth and fifth decades of life, predominantly in women. Skin manifestations are common (50% of cases) and clinically polymorphic. The nasal sinus tract is usually affected, and sometimes only one sign is observed. Kidney failure is common and can be life-threatening.
Macroscopy
The most common clinical presentation of GPA is palpable and non-palpable purpura of the lower extremities, which can progress to necrosis with the formation of vesicles, blisters and ulcers. Other possible cutaneous manifestations include subcutaneous nodules, livedo reticularis, infarcts (gangrene), pyoderma gangrenous ulcers, and urticarial plaques (Figure 3A). Oral lesions are usually characterized by nonspecific erosive or ulcerative lesions and, less commonly, gingival hyperplasia. Skin lesions follow a chronic, relapsing course, with episodes lasting from several weeks to several months. A rare form of GPA is limited to the facial mucosa and affects the upper respiratory tract, leading to the destruction of cartilage and bone. Figure 3. A) Pyoderma gangrenous ulcer on the right shoulder of a patient suffering from granulomatosis with polyangititis; B) necrotizing vasculitis with partial thrombosis, fibrinoid degeneration and neutrophil infiltration by leukocytoclasis of a medium-sized vessel at the dermal-subcutaneous junction (courtesy of Prof. F. Rongioletti).
Microscopy
Histological features depend on the type of biopsy of clinical lesions. Palpable and non-palpable purpura usually represents a typical aspect of LCV. In more aggressive skin lesions, there is a typical necrotizing granulomatous infiltrate of neutrophils, lymphocytes, plasma cells and rarely eosinophils involving small and medium-sized arteries and veins. Thrombi are often present, resulting in extensive necrosis (Figure 3B). The infiltrate may be perifollicular, and extravasation of red blood cells may be present.
Supporting research
DIF typically detects IgM and C3 deposits around small vessels in the dermis. Cytoplasmic (c) - ANCA against leukocyte proteinase 3 (PR3) is usually positive.
Eosinophilic granulomatosis with polyangitis (Churg-Strauss syndrome)
Eosinophilic granulomatosis with polyangititis (EGPA) is a rare systemic small vessel vasculitis. It typically begins with asthma, often associated with peripheral and tissue eosinophilia, and then progresses to necrotizing granulomatous vasculitis affecting almost every organ system. Renal involvement occurs in approximately 25% of patients. Also possible: eosinophilic gastroenteritis, peripheral neuropathy and cardiac involvement. It is most often observed in men in the third and fourth decades of life.
Macroscopy
Common findings include palpable purpura, petechiae, bruising, and cutaneous and subcutaneous nodules distributed symmetrically over the scalp and extremities (Figure 4A). Uriticaria, erythematous macules, multiple forms of erythema, and livedo pampiniformes are less common.
Figure 4. A) Erytumatous necrotic papules and plaques surrounded by a purple halo on the legs of a patient with eosinophilic granulomatosis with polyangitis; B) leukocytoclastic vasculitis around a small vessel associated with a granulomatous inflammatory infiltrate with numerous eosinophils (courtesy of Prof. R. Gianotti).
Microscopy
EGPA is characterized by LCV of small vessels (both postcapillary venules and arterioles) associated with the presence of large numbers of eosinophils in the dermis. Another typical feature is palaging granulomatous inflammation with necrosis, numerous eosinophils, and degenerative sheathed collagen bundles (red granulomas) within the vessels, surrounding dermis, and subcutaneous fat (Figure 4B).
Supporting research
Peripheral eosinophilia is helpful for diagnosis. DIF can detect IgM and C3 deposits in dermal vessels. Perinuclear (p)-ANCA antibodies against myeloperoxidase (MPO) are positive in two thirds of patients.
Medium vascular vasculitis
Polyarteritis nodosa
Polyarteritis nodosa (PAN) is a necrotizing disease of the small and medium sized muscular arteries. Since its first description, PAN has been divided into systemic and cutaneous forms. Systemic PAN is life-threatening and involves damage to multiple organs, including the kidneys, gastrointestinal tract, and nervous system. Cutaneous PAN has a benign course of chronic recurrence but no evidence of systemic disease. Extracutaneous manifestations include fever, myalgia, arthralgia, and sensory polyneuropathy. The pathogenesis of PAN is unknown, but many infectious agents (especially hepatitis B virus) are also involved in the formation of immune complexes.
Macroscopy
In cutaneous PAN, typical lesions have several pink to violet-red nodules primarily on the distal lower extremities. Lesions may appear on the surface of the livedo reticularis. In systemic PAN, the most common cutaneous findings are palpable purpura and subcutaneous nodules on the lower extremities. Livedo reticularis may also occur.
Microscopy
Early lesions show segmental necrotizing vasculitis of medium (or small) arterioles in the deep dermis. There is a mixed infiltrate with a predominance of neutrophils and noticeable thickening of the vessel wall. The eosinophilic ring of fibrinoid necrosis creates a target-like pattern. Leukocytoclasia is sometimes present. In older lesions, lymphocytes predominate.
Supporting research
DIF shows vascular deposits of IgM and C3. ANCAs are usually negative.
Conclusions. Cutaneous vasculitis may be a disease of a single organ, limited to the skin, or may be a sign of a multisystem condition. The diagnosis of vasculitis may be similar in terms of clinical and pathological features between the two groups of cutaneous and systemic vasculitides. Biopsy is mandatory for diagnosis, but skin function, possible presence of systemic symptoms, histological aspects and laboratory data are also necessary to classify the type of vasculitis and select appropriate treatment. The typical histological appearance of the general group of cutaneous vasculitides is leukocytoclastic vasculitis of small vessels, but larger dermal vessels may also be involved. Extravascular histopathological findings such as an inflammatory infiltrate enriched in eosinophils and/or granulomatous aspects, as well as systemic symptoms or signs are key diagnostic clues, as cutaneous vasculitis should be considered a diagnosis of exclusion.
Diagnostics
It is necessary to try to diagnose vasculitis even in the presence of the very first signs. The disease is serious and dangerous and requires immediate treatment.
If you suspect an illness, you must perform a series of examinations and tests. Appointed:
- angiography;
- general blood and urine tests;
- ECHO-cardiography;
- blood biochemistry;
- Ultrasound of the heart, abdominal organs and kidneys;
- X-rays of light.
The problem is that it is very difficult to diagnose vasculitis in the early stages. But more striking signs of the disease and a reason to sound the alarm appear even when several organs are affected at once.
In severe and rapid progression of the disease, an additional biopsy is performed, followed by a detailed study of tissue samples.
Treatment of vasculitis
Treatment of vasculitis is prescribed depending on the type of organs affected and concomitant diseases. If the primary form of the disease appears against the background of an allergy, it usually disappears on its own and does not require additional measures.
If the source of the disease affects vital organs, such as the heart, brain, kidneys or lungs, then the patient is prescribed a course of intensive therapy.
To stabilize blood circulation, treatment can be carried out both at home and in a hospital setting. Pregnant women, children and patients with severe forms of the disease are required to be hospitalized.
Use:
- chemotherapy;
- corticosteroids;
- NSAIDs;
- anticoagulants;
- enterosorbents;
- antiplatelet agents;
- antihistamines;
- cytostatics.
Additionally, plasmapheresis, hemosorption and immunosorption are included.
FAQ
What is the prognosis for vasculitis?
With timely consultation with a doctor, patient survival is 90%.
Is it possible to treat vasculitis at home?
If the patient’s condition and the identified stage of disease development allows this, then it is possible. Only the most difficult cases are hospitalized. Others can take medications on their own.
If you suspect vasculitis, who should you turn to for help?
First of all, you should make an appointment with a therapist, who, after collecting an anamnesis, will refer the patient to specialized specialists: a vascular surgeon, cardiologist, hematologist, etc.
Prevention
Unfortunately, it is impossible to protect yourself one hundred percent from the appearance of vasculitis. Preventive measures include maintaining immunity, a healthy lifestyle, and paying attention to health.
If a person has already been diagnosed with vasculitis, we must not forget that the disease can return, so you need to carefully monitor your well-being.
Medical specialists in St. Petersburg remind: the sooner treatment is started, the less negative impact the disease will have on the body. If symptoms appear, consult a doctor immediately. Registration by phone is carried out around the clock.