Sofia Kasatskaya “Nature” No. 2, 2016
about the authorSofya Alekseevna Kasatskaya — Junior Researcher, Laboratory of Genomics of Adaptive Immunity, Institute of Bioorganic Chemistry named after. Academicians M. M. Shemyakin and Yu. A. Ovchinnikov RAS. Area of scientific interests: T-cell immunity, neuro- and oncoimmunology. Winner of the “Bio/mol/text” competition 2015 in the category “Best article on immunology.” |
The body’s adequate defense response to the invasion of viruses, bacteria and other pathogens is to destroy the affected cells, preventing the spread of infection and the death of a large number of its own cells. If a cell infected with a virus notices it, innate immune processes are launched: autophagy (utilization of internal cell components using lysosome enzymes) and apoptosis (programmed cell death). However, there are a lot of pathogenic viruses and bacteria, and they are constantly changing beyond recognition. To cope with them, the adaptive immune system and its main participants - lymphocytes - are activated. The pinnacle of the evolution of adaptive immunity was the cytotoxic T-lymphocyte, or killer T-lymphocyte. To recognize a fragment of the virus (antigen) on an infected cell, it uses the T cell receptor
, TCR), randomly and independently assembled on each T cell in the thymus gland. The mechanism of TCR assembly is unique and is characteristic only of the immune system of vertebrates. It is believed that these advantages were first obtained by primitive fish about 500 million years ago, when, as a result of a retroviral infection, genes encoding special proteins (recombinases) responsible for the recombination of TCR genes were introduced into their gametes.
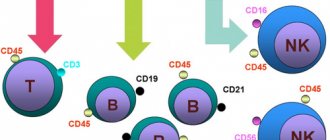
Classical human immunology is based on the study of immune cells in the blood simply because a blood sample can be taken from any patient and examined in normal and pathological conditions. It is on blood cells that the classification of T-lymphocytes was built - division into T-killers and T-helpers, which check the antigenic specificity of T-killers, give them a “license to kill” and are able to control the entire course of the immune response through soluble signaling molecules and cytokines. Later, a group of regulatory T cells was isolated from the T-helper branch, suppressing excess adaptive immunity.
But as yogurt commercials remind us, a significant portion of the immune system's cells are concentrated around the lining of the digestive tract and in other tissues. While there are about 6–15 billion T-lymphocytes in 5–6 liters of blood in an adult, there are 20 billion T-cells in the epidermis and skin [], and another 4 billion in the liver []. Is studying blood samples enough to fully characterize T cell function if there are more T cells in peripheral organs than in the bloodstream? And are classical subpopulations sufficient to describe all the types of T cells found in the human body?
Life cycle of a T lymphocyte
Each T cell, after TCR assembly, is tested for the functionality of the randomly assembled receptor (positive selection) and for lack of specificity for the body's own antigens, i.e. for the absence of an obvious autoimmune threat (negative selection). The stages of selection occur in the thymus gland; in this case, more than 90% of precursor cells die, failing to correctly assemble the receptor or undergo selective selection. The surviving T cells proliferate and exit the thymus into the bloodstream - these are naïve T lymphocytes that have not yet encountered the antigen. A naive T cell circulates in the blood and periodically enters the lymph nodes, where in the T-cell zone it contacts specialized cells that present it with a foreign antigen.
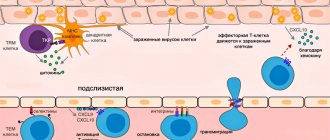
Effector T cell migration into tissue during viral infection []. Inflammatory signals from infected epithelial cells, with the participation of resident cells, are transmitted to the vascular endothelium, whose cells attract effector T cells with the chemokines CXCL9, CXCL10. Rolling:
When moving along the postcapillary venule in the tissue, the effector cell slows down, forming temporary contacts with E-selectins and P-selectins on endothelial cells.
Arrest:
The effector cell attaches tightly to the endothelium when LFA-1 and other alpha integrins interact with ICAM-1/VCAM-1/MAdCAM-1 (on the endothelium).
Transmigration:
effector T cell binds endothelial JAM-1 with molecules PECAM, CD99, LFA-1 and penetrates through endothelial cells into the submucosa
After encountering an antigen in the lymph node, the T cell acquires the ability to divide again - it becomes the precursor of memory T cells ( Stem Cell Memory T cells
, TSCM).
Among its descendants, central memory cells ( Central Memory T cells
, TCM) and effector progenitor cells (
Effector Memory T cells
, TEM) appear, which, when divided, give rise to short-lived effector cells that carry out the immune response (TEMRA cells) []. All these cells leave the lymph node and travel through the blood. The effector cells can then leave the bloodstream to carry out an immune response in the peripheral tissue of the organ where the pathogen resides. What then - another journey through the blood and lymph nodes?
Scheme of transition of descendants of activated T-lymphocytes between populations []. Explanations in the text
Stroma cells, i.e. the bases of the lymph node secrete signaling substances (chemokines) in order to invite the T cell to the lymph node. Lymph node chemokines are recognized by homing receptors CCR7 and CD62L. But both receptors are absent on effector cells. Because of this, it has long been a mystery how effector cells can get from peripheral tissue back to the secondary lymphoid organs - the spleen and lymph nodes.
Difficult choice of effector cell.
To home
is the process of homing, or migration of T cells, for example, to the place most familiar to naive cells - the lymph node. The alternative is not to travel through the body and become a resident tissue cell.
At the same time, data began to accumulate (on differences in TCR repertoires and transcription profiles between TEMs in the blood and in other tissues) that did not fit into the concept of constant migration of T cells between tissues and blood. It was decided to isolate a new subpopulation - resident memory cells ( Resident Memory T cells
, TRM), which inhabit a specific organ and do not recycle [].
Where do tissue resident cells first appear? These are the descendants of effector cells that have lost their ability to recycle. Some tissues peripheral to the immune system, such as the mucosa of the small intestine and the abdominal cavity, allow effector T lymphocytes to penetrate freely, while others allow very limited penetration. A large influx of effector T cells into these tissues is observed only during an inflammatory response. The second type of tissue includes the brain and spinal cord, separated by a barrier from the immune system, as well as many other tissues: peripheral ganglia, mucous membranes of the genitals and intestines, lungs, epidermis, eyes. The difference between the two tissue types is the expression of additional homing molecules for effector T cells, such as the adhesion molecule MadCAM-1 for penetration into the epithelium [].
Resident T cells in aging human tissues
A map of the relationships between the presence of individual subpopulations of T cells in different human organs, oddly enough, was compiled only in 2014. Donna Farber's team from Columbia University Medical Center in New York compared the phenotypes of T cells isolated from the blood and tissues of organ donors of all age groups from 3 to 73 years (total 56 donors) []. Analysis of T cell subpopulations using flow cytometry confirmed many data obtained using methods with lower resolution and lower statistics, and some features of the description of the immune system transferred from mouse immunology to humans, for example, a decrease in the content of naive T lymphocytes in all organs during aging of the body .
The decline in the number of naive T cells with age is associated with the rapid aging of the thymus, in which future T cells undergo the stages of TCR assembly, testing its functionality, and selection for lack of autoimmune potential. It is important not only to reduce the absolute number of naïve T cells, but also to reduce the diversity of the T-cell receptor repertoire, and therefore the ability to form an adaptive immune response to a previously unfamiliar infection []. For naive killer T cells, a progressive decline in abundance in the blood and lymph nodes was confirmed, although for naive helper T cells the negative correlation of abundance with age in this study was significant only for secondary lymphoid organs, but not for the blood.
Pathways of circulation of T-lymphocytes of various subpopulations []. Naive T cells, together with a subpopulation of TCM, travel through blood vessels and enter the T-cell zone of various lymph nodes; they do not enter the tissues, although they are found in their capillaries ( red trajectory
).
Effector TEM cells move through the lymph and bloodstream, can enter the lymph node, but do not enter the T-cell zone ( purple trajectory
).
Resident TRM cells (shown in green
in the skin and
different colors
in the mucous membranes) move only within the tissue (
green trajectory
)
Isolation of memory T lymphocytes, memory effector cells and short-lived effector cells from the mucous membranes of the lungs, small and large intestines, inguinal and mesenteric lymph nodes of organ donors made it possible for the first time to assess the dynamics of these populations in human tissues during aging. The proportion of central memory cells expectedly increases over the course of life, in accordance with the increase in the number of infections that the body has encountered and entered the memory library of the immune system. TEM effector memory cells rapidly fill the T cell niche in the child's tissues, quickly displacing naïve T cells by about 12 years of age. The number of terminally differentiated effector T-killer cells (TEMRA) also increases, but only in the lymph nodes and spleen; in nonlymphoid tissues, TEMRA abundance decreases. Short-lived terminally differentiated killer T cells are most often found in the blood, spleen and mucous membranes of the lungs at any age, but among T helper cells this subpopulation is represented by a vanishingly small number of cells. Similarly, there are few central memory cells among T-killers; they are predominantly located in the mucous membranes of two barrier tissues: the lungs and intestines.
In broad strokes, the distribution map of human T-lymphocytes can be outlined as follows: naïve T-cells travel through the blood and periodically enter secondary lymphoid organs; TEMRA killers are found in the blood, spleen and lungs. Apparently, central memory cells are characterized by a more individual distribution across tissues than other subpopulations: in any case, patterns of dynamics during aging of different tissues could not be identified. Effector memory cells, including the TRM subpopulation, dominate among T cells of mucosal barrier tissues. In general, with aging of T-cell immunity, non-lymphoid tissues show greater stability of subpopulations, while lymphoid tissues show greater age-related dynamics of T-cell types []. The stability of tissue cells is easier to explain if we understand which of the TEM effector cells remain in the tissue, become resident TRMs, and what events their life consists of after they stop traveling throughout the body.
References
- Litvinova, L.S., Gutsol, A.A., Sokhonevich N.A. and others. Basic surface markers of the functional activity of T-lymphocytes. Medical immunology, 2014. - No. 1.
- Histology (introduction to pathology) / ed. E.G. Ulumbekova, Yu.A. Chelysheva, 1997. - P. 15-17, 532 -546.
- Encyclopedia of clinical laboratory tests / ed. WELL. Titsa. - M.: Labinform, 1997. - P. 214-215.
- Saraiva, D., Jacinto, A., Borralho, P. et al. HLA-DR in Cytotoxic T Lymphocytes Predicts Breast Cancer Patients' Response to Neoadjuvant Chemotherapy. Front Immunol., 2021. - Vol. 9. - P. 2605.
How to distinguish resident tissue cells from admixtures of blood cells?
Resident T cells can be correctly, but inconveniently, determined each time by the ability of an individual cell to migrate to lymph nodes, so it is necessary to compile a list of characteristic features that can be used to identify membership in this subpopulation. Resident T lymphocytes in tissues that are the body's natural barriers (for example, in the lungs and small intestinal mucosa) are a bit like classical effector blood cells: they express the activated cell marker CD69, and the expression is stable throughout life during adulthood and aging and is characteristic of all non-lymphoid tissues. But in addition, CD69 colocalizes with the marker CD103, which denotes a group of adhesion molecules - integrins, which promote the attachment of resident T cells to the epithelium and to fibroblasts in the submucosa of the selected organ. For effector T cells in secondary lymphoid organs, the expression of CD103 integrins is completely uncharacteristic: TEM cells constantly maintain a motile phenotype.
The map compiled by Donna Farber's team has a major flaw: it is unclear how cleanly T-lymphocytes can be isolated from an organ, and what proportion of the analyzed cells are actually blood T-lymphocytes from capillaries inside the organ.
The issue of contamination by blood cells is especially acute for the lungs - it is no coincidence that the subpopulation composition of T cells in the lungs is unexpectedly similar to T cells in the blood and lymph nodes. The issue of blood cell contamination was elegantly solved for mouse T cells: experimental animals were infected with lymphocytic choriomeningitis virus after transplantation of a transgenic P14 T cell clone specific for this virus. As a result, during infection, the majority of circulating cells were represented by the virus-specific P14 clone, and its presence in tissues could be detected using fluorescent antibodies to TCR P14. Mice were injected into the blood with an anti-CD8 antibody to a marker of T-killer cells; it quickly spread through the bloodstream and bound to all T-killer cells in the blood (but not in tissues). By microscopy of organ sections, it was easy to distinguish resident killer TRMs from cells only recently released from the blood into the organ, labeled with anti-CD8 antibody []. The number of resident cells calculated by this method was 70 times higher than the number determined by flow cytometry; a difference of less than twofold was observed only for resident cells of the lymph nodes and spleen. It turns out that standard methods for isolating lymphocytes from organs are poorly suited for the analysis of killer resident cells and significantly underestimate the size of the population.
What can the study of Trm give to medicine?
Understanding how resident T cells work is absolutely essential for fighting infections that do not enter the bloodstream directly, but enter the body through barrier tissues—that is, for the vast majority of infections. Rational design of vaccines for protection against this group of infections can be aimed specifically at enhancing the first stage of protection with the help of resident cells: a situation in which optimally activated antigen-specific cells eliminate the pathogen in the barrier tissue is much more beneficial than triggering acute inflammation to call T lymphocytes from the blood, since there is less tissue damage.
The T-cell receptor repertoire of cells associated with mucosal barrier tissues is considered to be partially degenerate and public, that is, identical for many individuals in a population. However, biases in the isolation of T cells from organs, data bias resulting from the selection of only certain Caucasian donors into cohorts, and the overall small amount of accumulated sequencing data do not provide confidence in the public availability of T-cell receptor repertoires of TRM cells. While this would be convenient, vaccine design could be reduced to finding and modifying the most affinity and immunogenic peptides from a pathogen to interact with one of the publicly available TCR variants in the pathogen's barrier tissue.
Of course, understanding what T-cell receptors TRM cells carry on their surface is not enough to effectively manipulate immune responses in tissue. It is necessary to study in detail the factors influencing the colonization of tissues by certain T-cell clones and to understand the mechanisms of activation of local tissue immunity and induction of TRM tolerance. How are the niches of T-lymphocytes populated in the mucous membranes of a child before encountering a large number of pathogens and, accordingly, before the formation of a significant pool of effector memory T-cells - precursors of resident cells and central memory cells? Why and how, instead of the classical activation of lymphocytes, ignoring and tolerance reaction to microbes of non-pathogenic mucosal flora is formed? These questions are on the agenda in the study of resident cells of the immune system.
Determining the patterns of T-lymphocyte homing into specific tissues may provide an advantage in cellular immunotherapy of tumor diseases. Theoretically, killer T cells of the required tumor antigen specificity, activated in vitro, should kill the patient's tumor cells. In practice, such immunotherapy is complicated by the fact that tumor cells are able to suppress immune responses and render T-killer cells approaching the tumor into an inactive state of anergy. Often, anergic T-lymphocytes, primarily the TRM of a given tissue, accumulate in and around a growing tumor. Of the many active tumor-specific T cells injected into a patient, few will reach the target, and even these may be virtually useless in the immunosuppressive tumor microenvironment.
Deciphering the mechanisms that drive specific T cell clones to specific tissues could allow laboratory-engineered T cells to be targeted more effectively to tumors and usher in the era of accessible, personalized immunotherapy.
The work of resident T cells: do not confuse tourism with emigration
In a normal situation, mouse resident tissue cells hardly move within non-lymphoid tissue and are quite firmly attached by adhesion molecules to the stroma of the organ. When resident macrophages of the same tissue initiate an inflammatory response by secreting cytokines, TRMs become more motile and patrol the nearby epithelium in search of infected cells.
If the inflammatory reaction intensifies, then the cells understand this as a signal for reinforcement: TCM and TEM cells newly arriving from the blood are connected to the work of patrol TRM. These blood cells are much more mobile and move better in the epithelium. Does this mean that it is in the blood that killer T-cells are ready to act among TEM, and CD8+ TRMs perform helper and regulatory functions in the tissue?
On the one hand, T-helpers are more tissue-specific in the spectrum of T-cell receptors, i.e. There is very little overlap between the TCR repertoires of cells taken from different tissues, while cells of the same killer T cell clone are found in different tissues among TEMs []. The range of functions and repertoire of antigen specificity of TRM remains to be studied, but TRM killers definitely have the ability to destroy infected tissue cells. Moreover, in a model of murine polyomavirus infection in brain tissue, the affinity of virus-specific T-cell receptors of resident killer cells is higher than that of virus-specific central memory cells [].
However, the size of the T-cell population depends not only on the specificity of the TCR for infections that previously occurred in a given organ, but also on the homeostatic proliferation of T-cells - the proliferation of more successful cells to fill the organ's capacity according to the number of T-lymphocytes. By using the markers CD28 and CD127 on the cell surface, it is possible to distinguish cells recently and long ago activated through the TCR from those that received only a homeostatic signal to proliferation from the growth factor IL-7. As tissue ages, homeostatic cell proliferation begins to dominate over the proliferation of TCR-activated cells.
NKT cells, a type of liver-resident cell found in other tissues, often function independently of T-cell receptors. They can be activated by NK cell receptors through recognition not of individual antigens, but of general molecular patterns of danger and tissue stress. When activated, CD8+ NKT cells release cytotoxic granules and lyse suspicious tissue cells, for example, single tumor cells and virus-infected cells that express and display stress molecules on the outer membrane. With aging, the tendency of TRM to be activated without the T cell receptor, through NK cell receptors or cytokine signals, can lead to erroneous lysis of tissue cells, insufficient control of chronically infected or degenerating areas of the epithelium.
Pathological manifestations associated with the work of resident T cells include organ-specific autoimmune syndromes and chronic tissue inflammation syndromes. Examples of chronic inflammation maintained by resident T lymphocytes are contact dermatitis and psoriasis, and the mechanism is the release of inflammatory factors IL-17 by resident killer T cells and IL-22 by resident dermal helper T cells. CD8+ effector T killer cells located in the brain are similar in their set of membrane marker molecules to the TRM of the skin, intestines and lungs and are able to push the development of intermittent multiple sclerosis with periodic releases of inflammatory cytokines. It is unclear, however, whether there is a normal TRM population in the brain or whether these are T lymphocytes remaining in the tissue after a neurotropic viral infection [].
The functions of resident memory cells normally—in the absence of infection or chronic inflammation—may include cross-talk
(mutual regulation mainly through the secretion of cytokines and costimulatory molecules) with non-classical, poorly studied lymphoid cells.
They may be mucosal-associated γ/δ T cells, which carry an alternative variant of T-cell receptor assembly, or Innate Lymphoid
(ILCs), which share common features of the epigenetic landscape with T and B lymphocytes, but do not have T-/B- or NK-cell receptors [, ].
Proposed functions of tissue resident T-lymphocytes. Some functions can be performed in interaction with resident macrophages
TRM cells come into contact with antigen-presenting tissue cells—dendritic cells of the skin and resident tissue macrophages. Resident myeloid cells in different tissues are differentiated and slightly similar to each other. For example, marginal zone macrophages of the spleen, liver macrophages, and microglia (brain macrophages) will differ greatly in both morphology and range of functions. In addition to detecting antigens in tissue, resident macrophages are involved in regulating the processes of aging and tissue self-renewal, in particular, they secrete growth factors and cytokines that stimulate the division of tissue stem cells. In adipose tissue, for example, macrophages stimulate the differentiation of new fat cells, but when they enter an activated M1 state, they trigger inflammation and, instead of differentiating, cause existing fat cells to enlarge and swell. Concomitant changes in adipose tissue metabolism lead to the accumulation of fat mass and in recent years have been associated with the mechanisms of development of obesity and type II diabetes. In the skin, cytokines released by macrophages and resident γ/δ T cells stimulate stem cell division during epidermal and hair follicle stem cell regeneration [, ]. It can be assumed that helper TRM cells, when patrolling the epithelium and forming contacts with tissue macrophages, can modulate the spectrum and volume of growth factors secreted by the latter for stem cells, inflammatory cytokines and epithelial remodeling factors - and thereby participate in tissue renewal.